Perspective Oct 29 2020 Evaluating and Deploying Covid-19 Vaccines JL. Original Article from The New England Journal of Medicine Preliminary Findings of mRNA Covid-19 Vaccine Safety in Pregnant Persons.

Covid 19 Safety And Efficacy Of The Mrna 1273 Sarscov 2 Vaccine Moderna Against Sars Cov 2
In an ongoing multinational placebo-controlled observer-blinded pivotal efficacy trial we randomly assigned persons 16 years of.

Covid 19 vaccine new england journal of medicine. The New England Journal of Medicine March 3 at 900 AM In this video Paul Sax MD provides answers to questions about the Covid-19 vaccine specifically addressing how to answer questions from patients with a history of allergies those who are pregnant or breastfeeding and those who prefer to wait for herd immunity. Confirmed Covid-19 was defined according to the Food and Drug Administration FDA criteria as the presence of at least one of the following symptoms. Schwartz N Engl J Med 20203831703-1705.
Fever new or increased cough new or. The New England Journal of Medicine December 10 2020 Covid-19 has affected tens of millions of people globally since it was declared a pandemic by the WHO on March 11 2020. January 13 2021 -- Interim Phase 12a data were published today in the New England Journal of Medicine demonstrating that the Companys single-dose investigational COVID-19 vaccine candidate JNJ-78436735 being developed by the Janssen Pharmaceutical Companies of Johnson Johnson provided an immune response that lasted for at least 71 days the duration of time.
Original Article from The New England Journal of Medicine Effectiveness of Covid-19 Vaccines against the B16172 Delta Variant. The New England Journal of Medicine January 15 In this video Paul Sax MD a Professor of Medicine at Harvard Medical School and an infectious disease specialist provides answers. Many pregnant persons in the United States are receiving messenger RNA mRNA coronavirus disease 2019 Covid-19 vaccines but data are limited on their safety in pregnancy.
Severe acute respiratory syndrome coronavirus 2 SARS-CoV-2 infection and the resulting coronavirus disease 2019 Covid-19 have afflicted tens of millions of people in a worldwide pandemic. Johnson Johnson Single-Shot COVID-19 Vaccine Demonstrated a Durable Immune Response and Elicited Dual Mechanisms of Protection Against Delta and Other SARS-CoV-2 Variants of Concern in Data Published in New England Journal of Medicine. A study published in The New England Journal of Medicine on 16 March 2021 suggests that a two-dose regimen has an efficacy of only 104 95 CI 768 to 548 against mild to moderate COVID-19 infection in almost 1500 HIV-negative young adults.
Safe and effective vaccines are needed urgently. Phase 1 trial of two vaccine candidates in younger and older adults added to earlier interim safety and immunogenicity data regarding BNT162b1 in younger adults from trials in Germany and the United States support the. NEW BRUNSWICK NJ April 21 2021 Johnson Johnson the Company today announced publication in the New England Journal of Medicine of primary data from the Phase 3 ENSEMBLE clinical trial for its single-dose COVID-19 vaccine developed by the Janssen Pharmaceutical Companies of Johnson Johnson Janssen.
New entities and investments have aided the pursuit of Covid-19 vaccines. Safety and Efficacy of a Single-Dose Vaccine against Covid-19 The New England Journal of Medicine June 12 at 900 AM Since its emergence in late 2019 the SARS-CoV-2 pandemic has caused high morbidity and mortality and new variants are spreading rapidly. Covid-19 Vaccine Frequently Asked Questions with Paul Sax MD.
Editorial from The New England Journal of Medicine mRNA Covid-19 Vaccines in Pregnant Women. Safe and effective vaccines are urgently needed. The safety and immunogenicity data from this US.
New England Journal of Medicine study finds 82 miscarriage rate among women vaccinated against covid-19. Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates. The results of this trial provide further evidence that immunization with a protein-based adjuvanted vaccine such as NVX-CoV2373 can prevent Covid-19 caused by either B117 or non.
From December 14 2020 to February 28 2021 we used data from the v-safe after vaccination health checker surveillance system the v-safe pregnancy registry and the Vaccine.

Developing Vaccines For Covid 19 A Summary Of An Opinion Article Published In The New England Journal Of Medicine

Covid 19 What Does Vaccine Efficacy Really Mean Diu News

Lancet Nejm Retract Covid 19 Studies That Sparked Backlash

What Are Your Chances Of Catching Covid 19 After Getting Vaccinated A New Study Measures That Risk Cbs 17
Progress Reported On One Dose J J Vaccine Covid 19 Reinfections Seen As Rare Reuters
The New England Journal Of Medicine Quick Take A Covid 19 Mrna Vaccine In Older Adults Facebook
Moderna Pfizer Covid 19 Vaccines May Reduce Coronavirus Transmission Science News

Sinovac S Covid 19 Vaccine Is Able To Prevent Over 80 Of Deaths According To Turkish And Chilean Studies Health Policy Watch
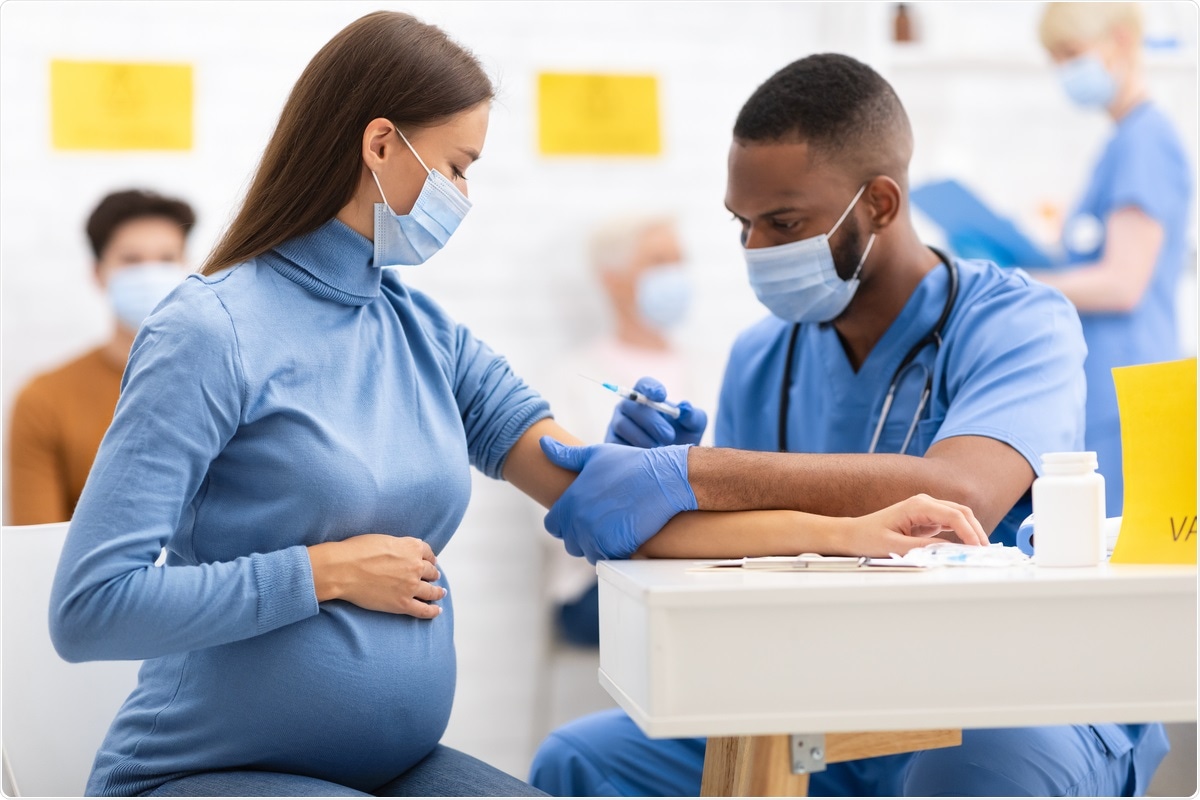
Study Reports On Safety Of Covid 19 Mrna Vaccines In Pregnancy

Stephen J Thomas Md Co Authors Paper Published In New England Journal Of Medicine About Pfizer S Covid 19 Vaccine Upstate News Suny Upstate Medical University

Safety And Efficacy Of Single Dose Ad26 Cov2 S Vaccine Against Covid 19 Nejm

Covid 19 Vaccine Frequently Asked Questions Hiv And Id Observations

Coronavirus Vaccines May Have Side Effects But That Should Not Be Any Deterrent Coronavirus Outbreak News

Baseless Claims About Mrna Vaccines And Miscarriages

Covid 19 Vaccine Pfizer Trial Lacks Asian Data

Safety And Efficacy Of Single Dose Ad26 Cov2 S Vaccine Against Covid 19 Nejm
The New England Journal Of Medicine Posts Facebook

Safety And Efficacy Of Single Dose Ad26 Cov2 S Vaccine Against Covid 19 Nejm

Aerosol And Surface Stability Of Sars Cov 2 As Compared With Sars Cov 1 Nejm




0 Comments